Nigeria’s drug regulatory agency has issued an urgent public health alerts warning that counterfeit versions of two critical cancer medicines are circulating in the country and that patients who use them risk treatment failure, disease progression and death.
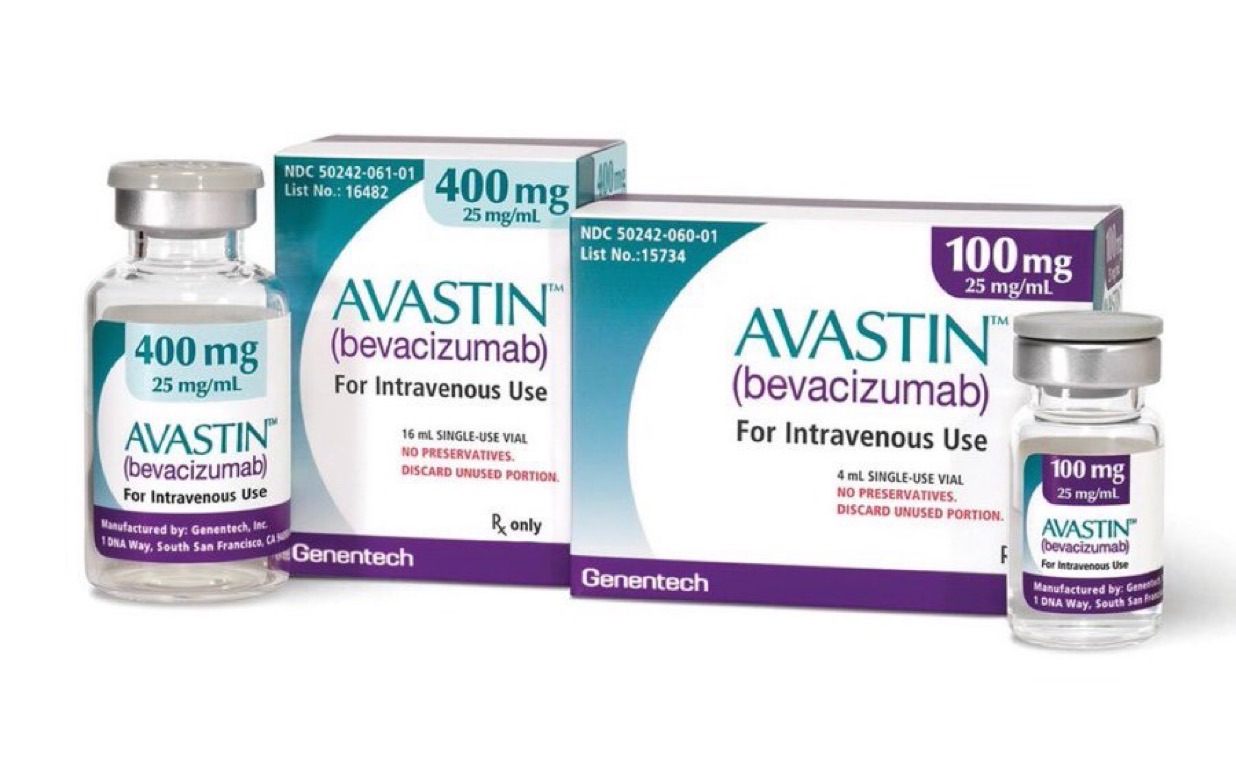
The National Agency for Food and Drug Administration and Control has confirmed the presence of counterfeit batches of Avastin 400mg/16ml and Tecentriq 1200mg/20ml in Nigeria’s pharmaceutical supply chain, following official communication from Roche Nigeria which reported receiving multiple complaints from healthcare professionals across the country.
The drugs being faked are not minor medications. Avastin is a vital therapy used to treat brain tumours in adults, while Tecentriq is a specialised immunotherapy used for lung, liver and skin cancers. Patients battling these advanced and life threatening conditions are now unknowingly at risk of receiving drugs that may contain no active ingredients at all or worse, harmful substances.
The counterfeit drugs were reportedly sold at significantly lower prices ranging between N180,000 and N350,000 far below the cost of genuine versions with patients in several instances bringing the suspicious products directly to healthcare facilities after purchasing them from unauthorised sources. That price gap is the clearest warning sign, and the one that has already lured vulnerable patients into danger.
The counterfeit products are identifiable to trained eyes. Investigations revealed multiple red flags including batch numbers that do not exist in the Roche manufacturer’s database, poor quality printing and spelling errors, incorrect placement of information on packaging, tamper evident labels inconsistent with genuine products and invalid serial numbers.
“The batch number does not exist in the Roche database, making lot tracing impossible,” NAFDAC noted.
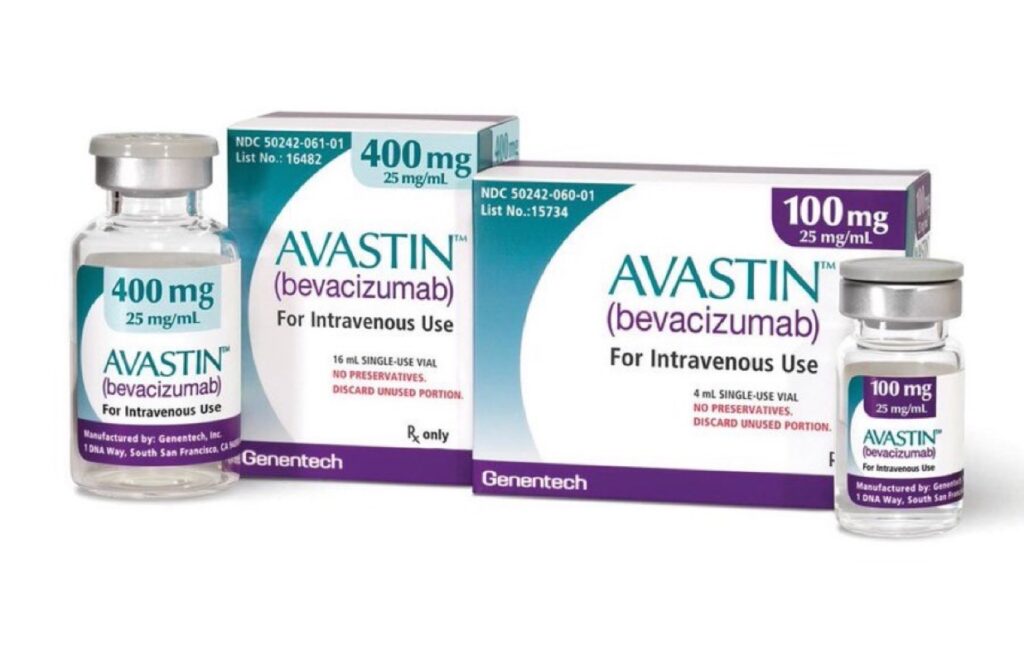
The specific counterfeit batches identified are Avastin 400mg/16ml with batch numbers H4239A70, H2290A34 and A3508B02, as well as Tecentriq 1200mg/20ml with batch number B3071A12.
Any product carrying these batch numbers should be treated as counterfeit and not administered to patients.
There is however one important caveat in NAFDAC’s own alert that deserves transparency. Chemical analysis of the suspected products could not be carried out as the investigation depended on packaging photographs sent by complainants and no physical samples were available.
The confirmation is therefore based on packaging evidence rather than laboratory analysis which means the exact contents of the fake vials remain unknown, making them potentially even more dangerous.
NAFDAC has directed all zonal directors and state coordinators to carry out immediate surveillance and mop up the counterfeit products across every zone and state in the country.
Importers, distributors, retailers, healthcare professionals and caregivers have been advised to exercise caution and vigilance within the supply chain to avoid the importation, distribution, sale and use of the counterfeit products.
Members of the public are encouraged to report suspected counterfeit or substandard medicines through NAFDAC offices, its hotline, or via email.
Nigeria’s drug regulatory agency has issued an urgent public health alerts warning that counterfeit versions of two critical cancer medicines are circulating in the country and that patients who use them risk treatment failure, disease progression and death.
The National Agency for Food and Drug Administration and Control has confirmed the presence of counterfeit batches of Avastin 400mg/16ml and Tecentriq 1200mg/20ml in Nigeria’s pharmaceutical supply chain, following official communication from Roche Nigeria which reported receiving multiple complaints from healthcare professionals across the country.
The drugs being faked are not minor medications. Avastin is a vital therapy used to treat brain tumours in adults, while Tecentriq is a specialised immunotherapy used for lung, liver and skin cancers. Patients battling these advanced and life threatening conditions are now unknowingly at risk of receiving drugs that may contain no active ingredients at all or worse, harmful substances.
The counterfeit drugs were reportedly sold at significantly lower prices ranging between N180,000 and N350,000 far below the cost of genuine versions with patients in several instances bringing the suspicious products directly to healthcare facilities after purchasing them from unauthorised sources. That price gap is the clearest warning sign, and the one that has already lured vulnerable patients into danger.
The counterfeit products are identifiable to trained eyes. Investigations revealed multiple red flags including batch numbers that do not exist in the Roche manufacturer’s database, poor quality printing and spelling errors, incorrect placement of information on packaging, tamper evident labels inconsistent with genuine products and invalid serial numbers.
“The batch number does not exist in the Roche database, making lot tracing impossible,” NAFDAC noted.
The specific counterfeit batches identified are Avastin 400mg/16ml with batch numbers H4239A70, H2290A34 and A3508B02, as well as Tecentriq 1200mg/20ml with batch number B3071A12.
Any product carrying these batch numbers should be treated as counterfeit and not administered to patients.
There is however one important caveat in NAFDAC’s own alert that deserves transparency. Chemical analysis of the suspected products could not be carried out as the investigation depended on packaging photographs sent by complainants and no physical samples were available.
The confirmation is therefore based on packaging evidence rather than laboratory analysis which means the exact contents of the fake vials remain unknown, making them potentially even more dangerous.
NAFDAC has directed all zonal directors and state coordinators to carry out immediate surveillance and mop up the counterfeit products across every zone and state in the country.
Importers, distributors, retailers, healthcare professionals and caregivers have been advised to exercise caution and vigilance within the supply chain to avoid the importation, distribution, sale and use of the counterfeit products.
Members of the public are encouraged to report suspected counterfeit or substandard medicines through NAFDAC offices, its hotline, or via email.